Scientists uncover the earliest steps of protein folding in human cells
Four research groups joined forces to reveal the very first moments of a protein’s life in human cells. They found that as soon as an amino acid chain exits from the ribosome, a protein called NAC binds and folds it into the correct structure. This finding fundamentally rewrites our textbook knowledge on how human cells suppress diseases caused by protein folding errors. The researchers published their findings in the journal Molecular Cell on March 23.
Although the NAC protein and its binding to the ribosome have been known for a long time, its core function has remained elusive. Due to the dynamics of the involved molecules and their fundamental role in cells, this problem could not be resolved using a single method. Hence, the research organizations Heidelberg University, ETH Zurich, Caltech, and AMOLF teamed up to try a different approach. By bringing their different techniques to the table, they were able to put all the pieces of the puzzle together.
Three unique approaches
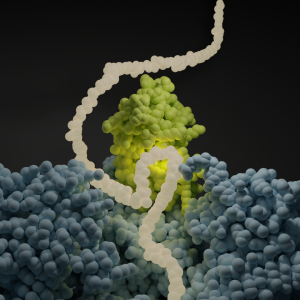
It started at Heidelberg University. Under supervision of Bernd Bukau and Guenter Kramer, a method was developed to determine where NAC binds on the amino acid chains. The team exploited the fact that ribosomes must read the genetic code stored in our DNA to make proteins. By sequencing (determining) the parts of the genetic code that the ribosomes were reading when NAC was bound, they could identify those NAC binding sites. This revealed that NAC binds to a large fraction of all proteins in the cell, and also at which parts.
A second group of researchers looked at the mechanisms at play. How does the interaction between NAC and the newly created proteins take place? And what does that binding do to the protein? The AMOLF team led by Sander Tans tackled this challenge with the group of Shu-Ou Shan at Caltech. Using a range of single molecule methods, such as fluorescence and optical tweezers, they studied how NAC moves from the ribosome to the amino acid chain, while monitoring the compactness of that chain. They could only do so because of the Heidelberg results, as they showed which proteins to focus on. These single-molecule experiments revealed the function of NAC – that it induces folding of the amino acid chain into a functional protein.
The third type of experiment at ETH Zurich zoomed in to the smallest – near atomic – level. Under supervision of Nenad Ban, the team used a cryo-electron microscope to take a ‘picture’ of NAC as it sits on the ribosome. By freezing the molecules quickly, the researchers were able to preserve the natural structure. The research team discovered the exact location on the NAC protein responsible for binding the amino acid chains. This finding allowed them to make a NAC variant using mutations, which is non-sticky at this binding location. These variants could indeed no longer fold proteins, thus beautifully confirming the mechanism.
Combining strengths to reveal the full picture
The three approaches are unique and highly advanced, yet each has its limitations. The Heidelberg method was able to determine where NAC binds and on which proteins, but they could not catch the effect of this binding. These movement and folding dynamics could be measured by the AMOLF and Caltech teams, but their methods in turn could not ‘see’ what this interaction between the ribosome, NAC, and synthesized protein looks like in detail. What these teams could not contribute was picked up by the ETH Zurich team with their cryo-electron microscope technique.
This collaboration began in 2022, when three of the four research organizations received an ERC Synergy grant to develop this combined approach. Although they have published several papers together since then, the NAC project is the first to bring everything together so effectively. It perfectly exemplifies the goal of a synergy grant: becoming stronger together. Enthusiastically, Sander Tans says, “It is beautiful to see how it all comes together and how these different techniques contribute to revealing truly essential information”. Bernd Bukau (Heidelberg University) adds: “This collaboration shows how important international networking is for the promotion of scientific progress”.
Learn more
- If you have questions about this research, contact Sander Tans at: s.tans@amolf.nl
- The paper ‘NAC promotes co-translational protein folding at the ribosomal tunnel exit’ was published in the journal Molecular Cell.
- Read full paper


